To assess the financial conflicts of interest (COI) reported by authors of major clinical practice guidelines (CPG) in Spain, based on their COI declaration.
MethodWe conducted a cross-sectional study comparing the financial COI declared by CPG authors with the transfers of value reported by pharmaceutical companies. Author declarations were extracted from CPG developed for the management of diverse disease conditions. Data on industry transfers of value was obtained from pharmaceutical company websites. The results are presented using descriptive statistics.
ResultsA total of 60 CPG were included, comprising 704 individual authors. As some individuals contributed to more than one CPG, analyses were conducted at the authorship level (n=810). Only 137 authorships (16.9%) provided accurate COI disclosures, while 430 (53.1%) were inaccurate or missing. In addition, 376 authorships (46.4%) lacked a COI declaration. Overall, more than three quarters of the authorships of CPG supported by five scientific societies were associated with unreliable COI declarations, despite 564 authors (80%) having received transfers of value from the pharmaceutical industry.
ConclusionsThis study assessed the accuracy of financial COI disclosures among authors of CPGs and identified gaps in transparency. Further research is needed to determine their implications on clinical recommendations and healthcare decision-making.
Analizar los conflictos de intereses (CDI) financieros reportados por los autores de las principales guías de práctica clínica (GPC) en España, a partir de sus declaraciones de CDI.
MétodoRealizamos un estudio transversal comparando los CDI financieros declarados por autores de GPC con las transferencias de valor reportadas por la industria farmacéutica. Las declaraciones de los autores se extrajeron de GPC desarrolladas para el manejo de diversas enfermedades. Los datos sobre las transferencias de valor de la industria se obtuvieron de las páginas web de las compañías farmacéuticas. Los resultados se presentan utilizando estadísticas descriptivas.
ResultadosSe incluyeron un total de 60 GPC, que comprendían 704 autores individuales. Dado que algunos autores contribuyeron a más de una GPC, los análisis se realizaron por autorías (n=810). Tan solo 137 autorías (16,9%) presentaron declaraciones de CDI precisas, mientras que 430 (53,1%) fueron inexactas o ausentes. Además, 376 autorías (46,4%) no presentaron declaración de CDI. En conjunto, más de tres cuartas partes de las autorías de las GPC avaladas por cinco sociedades científicas se asociaron con declaraciones de CDI poco confiables, pese a que 564 autores (80%) recibieron transferencias de valor de la industria farmacéutica.
ConclusionesEste estudio evaluó la precisión de las declaraciones de CDI financieros en autores de GPC e identificó deficiencias en la transparencia. Se requiere investigación adicional para determinar sus implicaciones en las recomendaciones clínicas y la toma de decisiones sanitarias.
Commercial determinants of health, defined as the set of systems, practices and pathways through which commercial actors influence health and equity,1 shape health and equity, with the pharmaceutical industry acting as a major actor through research, education, and clinical practice. Transparent conflict of interest (COI) disclosures are essential to safeguard the independence of science and medical decision-making.2 Industry payments to researchers and other transfers of value (TOV) can introduce bias into the generation and dissemination of evidence.3 Despite their importance, COI reporting often remains incomplete or inaccurate, highlighting ongoing gaps in transparency.4
The accuracy of interest declarations is critical in clinical practice guidelines (CPG), given their influence on clinical decision-making. Several studies have examined the frequency and quality of interest declarations in CPG. A 2019 systematic review5 involving more than 14,000 CPG authors, reported that 45% had at least one COI, and 32% had undisclosed industry payments. However, this study collected data mostly from USA guidelines. Similarly a North-American6 study of 14 hyperlipidemia and diabetes CPG found that 48% of panel members disclosed COI, while 11% of those reporting no interests were subsequently identified as having undisclosed COI. The study also observed a lower prevalence of COI among authors of government-sponsored guidelines, although COI identification relied on indirect methods, such as prior publications and online sources, which may have underestimated industry relationships.
With respect to scientific societies issuing CPG, Campsall et al.7 studied 95 organizations producing 290 CPG, demonstrating that 63% reported industry funding, organizations with stronger transparency policies issued fewer positive recommendations for patented biomedical products. Nevertheless, this study included only guidelines listed in the National Guideline Clearinghouse, limiting its applicability to other scientific societies, and did not assess COI at the individual author level. In Canada, another study reported that 75% of CPG included at least one author with declared financial COI, and in 54% of cases these relationships involved manufacturers of drugs recommended in the guidelines.8 However, the restricted national scope of this analysis limits its generalizability to European contexts.
The aggregate impact on population health from preventive or therapeutic clinical decisions for common health problems is of great magnitude,9,10 hence the relevance of ensuring that CPG are drafted through procedures that guarantee their independence from interests extraneous to population health. It is important to consider that recommendations regarding the use and interpretation of CPG include the question of the potential influence of COI,2,11 and it is known that financial COI are associated with favorable recommendation among guidelines and recommendation documents.12 If information on the quality of these declarations is unavailable, the recommendation is not applicable.
This study analyzes the prevalence of financial COI disclosures and its accuracy, comparing them with the TOV reported by pharmaceutical industry, made by the authors of major CPG in Spain.
MethodDesignWe conducted a cross-sectional study comparing COI declarations from Spanish CPG authors with records of TOV reported by pharmaceutical industries between 2016 and 2022.
Eligibility criteriaWe defined a financial COI as a TOV made by one of the selected companies to one of the selected authors in the previous three years before the publication of the CPG.
CPG were eligible for inclusion if they were published between 2017 and 2023, were developed by or officially endorsed by a Spanish scientific society or institution, and addressed the prevention, diagnosis or management of health conditions relevant to the Spanish population. The selection of target conditions was primarily guided by the Global Burden of Disease framework, using the 20 leading causes of disability-adjusted life years and mortality in Spain to ensure population-level relevance.13 Accidental causes and ill-defined categories (e.g., “other neuromuscular disorders” or “other tumors”) were excluded. Although Global Burden of Disease indicators provided the initial structure for selecting conditions, they were not applied as the sole criterion. Additional pragmatic considerations were incorporated to reflect the context of guideline development and implementation within the Spanish National Health System. These included the relevance of the guideline to public health priorities. Our intention was to identify guidelines addressing clinical practice events related to these 20 causes, even when some of the conditions were not, strictly speaking, specific causes of disease burden.
In cases where a Spanish scientific society did not issue a specific national guideline but explicitly referred to an international one (e.g., European Society of Cardiology), the same eligibility criteria were applied; however, only authors affiliated with Spanish institutions were included. The final selection of CPG was reviewed and agreed upon by all authors of the present study.
Information sources and search strategyEligible CPG and their authors were identified through a structured search of multiple sources. Guidelines were retrieved from the official websites and/or scientific journals of the corresponding Spanish scientific societies or institutions, as well from GuiaSalud (https://portal.guiasalud.es/, consulted in March 2023), a Spanish Ministry of Health platform that coordinates and endorses evidence-based CPG, pursuing quality, safety and equity of healthcare delivery in Spain. The search strategy was designed to capture guidelines addressing prevalent and high-impact conditions for which formal recommendations on prevention, diagnosis and/or management were likely to exist.
Data extraction and managementData were extracted manually from all included CPG (see Table S1 in online Supplementary Material). For each guideline, we recorded the title, year of publication, and sponsoring scientific society. For each listed author, we collected name, inferred gender, role in guideline development, and the presence and content of COI declarations, including explicit statements of no conflicts and cases in which no declaration was provided. Author gender and coordination roles were inferred from information reported in the CPG.
Financial COI were assessed by collecting data on TOV from the pharmaceutical industry. Pharmaceutical companies were selected based on sales volume, annual turnover,14 and the availability of publicly accessible disclosure documents (see Table S2 in online Supplementary Material). TOV data were obtained from the transparency sections of the websites of companies adhering to Farmaindustria's Code of Good Practices,15 which follows the European Federation of Pharmaceutical Industries and Associations reporting model.16 For each author, we identified TOV made within the three years preceding CPG publication, using records published between 2016 and 2022 when available. TOV were aggregated across the four general payment categories: registration fees, travel and accommodation, honoraria and related expenses.
Data extraction was performed by two authors, with discrepancies resolved by consensus. Authorships were classified according to the availability and content of COI declarations and then grouped into nine categories based on the correspondence between declared COI and identified TOV:
- •
Group 1: authorships not declaring COI but have received TOV prior to the CPG publication.
- •
Group 2: authorships not declaring COI and have not received TOV.
- •
Group 3: authorships not declaring COI and we just recorded TOV the same year or after the CPG publication.
- •
Group 4: authorships declaring absence of COI but have received TOV prior to the CPG publication.
- •
Group 5: authorships declaring absence of COI and have not received TOV.
- •
Group 6: authorships declaring absence of COI and we just recorded TOV the same year or after the CPG publication.
- •
Group 7: authorships declaring COI and declared all TOV received prior to the CPG publication. This group also includes authors who received no registered payments after our search.
- •
Group 8: authorships declaring COI but in a lower magnitude than our TOV records.
- •
Group 9: authorships declaring COI and we just recorded TOV the same year or after the CPG publication.
These were further summarized into four categories reflecting the reliability of COI declarations: reliable declarations (groups 2, 5 and 7), inaccurate or missing declarations (groups 1, 4 and 8), absent declarations with no payment records (group 2), and not verifiable declarations (groups 3, 6 and 9). This classification supported subsequent analyses comparing declared and industry-reported COI. Data management and processing were conducted using Microsoft Access.
The complete anonymized database can be consulted in: https://data.mendeley.com/datasets/xgbhfs629f/1.
Data analysisData were summarised using descriptive statistics. Categorical variables are presented as absolute frequencies and percentages, and continuous variables as medians with interquartile ranges (IQR). Analyses focused on describing authorship characteristics and the reliability of COI declarations. Specifically, we assessed the availability and content of COI disclosures, the distribution of authors across the nine predefined COI classification groups based on declared conflicts and recorded TOV, and their aggregation into broader reliability categories.
Comparisons were used to explore patterns and differences in authorship distributions across COI declaration and reliability categories, as well as the correspondence between declared COI and industry-reported TOV prior to guideline publication. Where appropriate, comparisons between categorical variables were assessed using Chi-square or Fisher's exact test; p values were 2-tailed, and statistical significance was set at less than 0.05. All analyses were conducted using Microsoft Excel (Microsoft Corporation, 2018).
ResultsWe selected 60 CPG that met our eligibility criteria (see Table S1 in online Supplementary Material), comprising 704 individual authors and 810 authorship instances; because some authors contributed to multiple guidelines, all 810 authorships were analyzed.
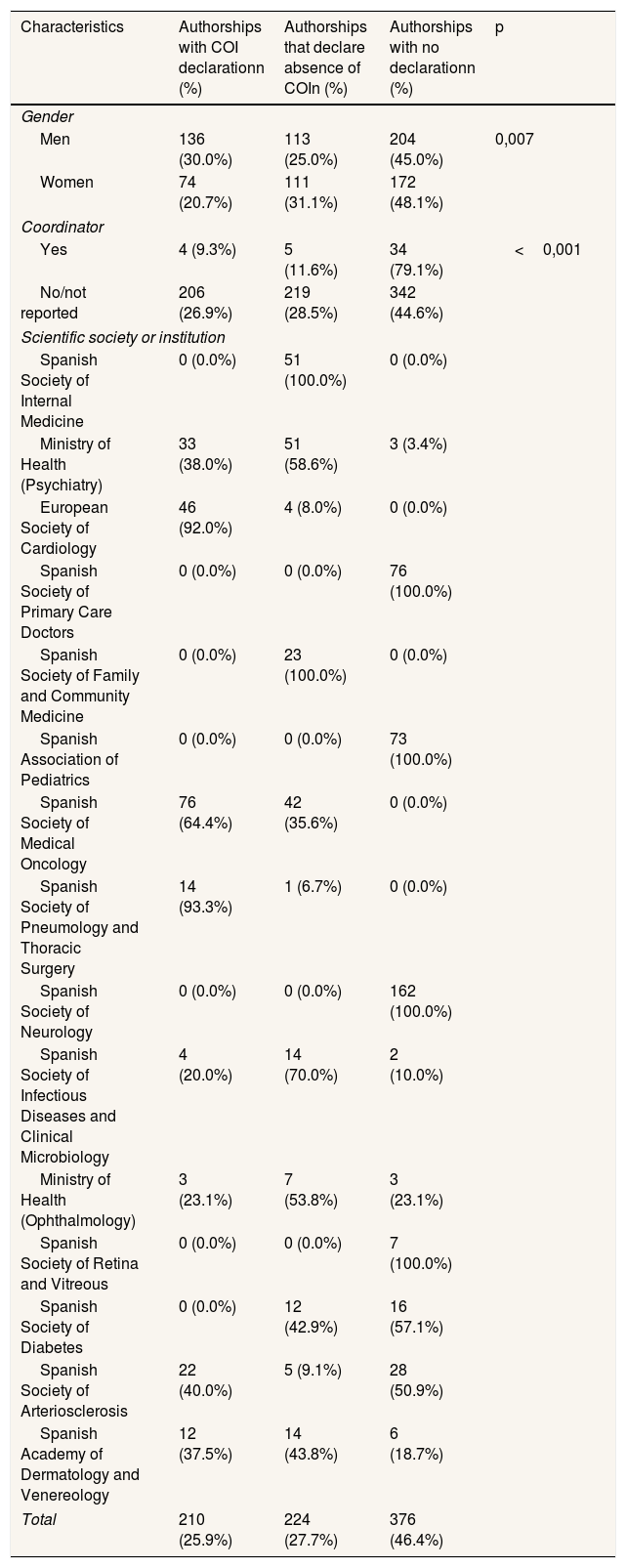
Regarding COI declarations, as shown in Table 1, more than half of the authorships (53.6%, n=434) included a COI statement, while the remaining cases (46.4%, n=376) had no available declaration. Of the declarations provided, 224 (51.6%) reported no COI, whereas 210 (48.4%) disclosed conflicts prior to the drafting of the CPG. Differences were observed between men and women authors (p=0.007), with men more frequently reporting receipt of payments than women, 30.2% and 20.7%, respectively.
Distribution of authorships by gender, coordination role, and scientific society according to the presence or absence of a conflict of interest declaration.
| Characteristics | Authorships with COI declarationn (%) | Authorships that declare absence of COIn (%) | Authorships with no declarationn (%) | p |
|---|---|---|---|---|
| Gender | ||||
| Men | 136 (30.0%) | 113 (25.0%) | 204 (45.0%) | 0,007 |
| Women | 74 (20.7%) | 111 (31.1%) | 172 (48.1%) | |
| Coordinator | ||||
| Yes | 4 (9.3%) | 5 (11.6%) | 34 (79.1%) | <0,001 |
| No/not reported | 206 (26.9%) | 219 (28.5%) | 342 (44.6%) | |
| Scientific society or institution | ||||
| Spanish Society of Internal Medicine | 0 (0.0%) | 51 (100.0%) | 0 (0.0%) | |
| Ministry of Health (Psychiatry) | 33 (38.0%) | 51 (58.6%) | 3 (3.4%) | |
| European Society of Cardiology | 46 (92.0%) | 4 (8.0%) | 0 (0.0%) | |
| Spanish Society of Primary Care Doctors | 0 (0.0%) | 0 (0.0%) | 76 (100.0%) | |
| Spanish Society of Family and Community Medicine | 0 (0.0%) | 23 (100.0%) | 0 (0.0%) | |
| Spanish Association of Pediatrics | 0 (0.0%) | 0 (0.0%) | 73 (100.0%) | |
| Spanish Society of Medical Oncology | 76 (64.4%) | 42 (35.6%) | 0 (0.0%) | |
| Spanish Society of Pneumology and Thoracic Surgery | 14 (93.3%) | 1 (6.7%) | 0 (0.0%) | |
| Spanish Society of Neurology | 0 (0.0%) | 0 (0.0%) | 162 (100.0%) | |
| Spanish Society of Infectious Diseases and Clinical Microbiology | 4 (20.0%) | 14 (70.0%) | 2 (10.0%) | |
| Ministry of Health (Ophthalmology) | 3 (23.1%) | 7 (53.8%) | 3 (23.1%) | |
| Spanish Society of Retina and Vitreous | 0 (0.0%) | 0 (0.0%) | 7 (100.0%) | |
| Spanish Society of Diabetes | 0 (0.0%) | 12 (42.9%) | 16 (57.1%) | |
| Spanish Society of Arteriosclerosis | 22 (40.0%) | 5 (9.1%) | 28 (50.9%) | |
| Spanish Academy of Dermatology and Venereology | 12 (37.5%) | 14 (43.8%) | 6 (18.7%) | |
| Total | 210 (25.9%) | 224 (27.7%) | 376 (46.4%) | |
COI: conflict of interest.
Guideline coordinators declaration of COI was lower than other contributors (79.1% of coordinators did not submit a declaration, compared to 44.6% among the remaining authors). Additionally, four scientific societies did not systematically include COI statements.
Regarding the 704 authors who participated in these CPG, 80.1% (n=564) received payments, amounting to 4788 transactions with a total value of 13,078,785.66 € (median per author: 7544.85 €; IQR: 23,597.20 €).
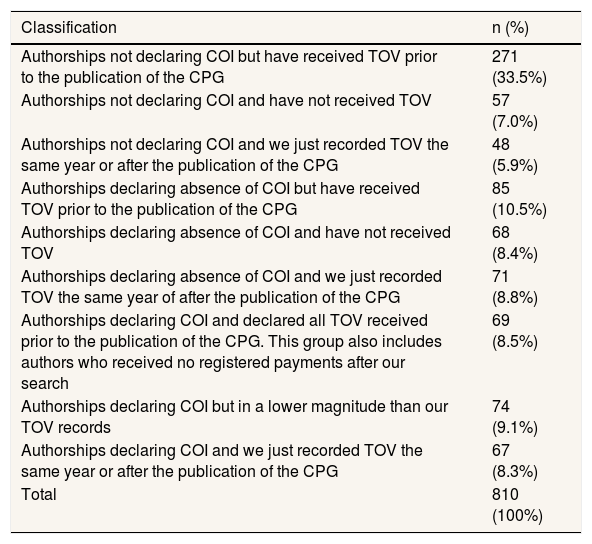
The accuracy of the COI declarations submitted by authors in the CPG was limited (Table 2). In up to 271 of the authorships (33.4%), no COI declaration was provided despite records indicating that the individual had received payments from pharmaceutical companies. Additionally, 85 authors (10.5%) declared no conflicts despite having received payments, and 74 (9%) disclosed COIs that were of a lower magnitude than those recorded in the official TOV. Only 68 of the authorships (8.4%) included a declaration of no COI that was fully consistent with the financial disclosures on record.
Distribution of authorships based on the presence of conflict of interest records and the declarations submitted.
| Classification | n (%) |
|---|---|
| Authorships not declaring COI but have received TOV prior to the publication of the CPG | 271 (33.5%) |
| Authorships not declaring COI and have not received TOV | 57 (7.0%) |
| Authorships not declaring COI and we just recorded TOV the same year or after the publication of the CPG | 48 (5.9%) |
| Authorships declaring absence of COI but have received TOV prior to the publication of the CPG | 85 (10.5%) |
| Authorships declaring absence of COI and have not received TOV | 68 (8.4%) |
| Authorships declaring absence of COI and we just recorded TOV the same year of after the publication of the CPG | 71 (8.8%) |
| Authorships declaring COI and declared all TOV received prior to the publication of the CPG. This group also includes authors who received no registered payments after our search | 69 (8.5%) |
| Authorships declaring COI but in a lower magnitude than our TOV records | 74 (9.1%) |
| Authorships declaring COI and we just recorded TOV the same year or after the publication of the CPG | 67 (8.3%) |
| Total | 810 (100%) |
COI: conflict of interest; CPG: clinical practice guidelines; TOV: transfer of value.
For 186 (22,96%) authorships, only records from the same year or after the publication of the CPG were available (groups 3, 6 and 9 in Table 2). This limits a reliable assessment of the accuracy of the COI declarations submitted by these authors. However, for the remaining 624 (77%) cases, it was possible to distinguish and classify declarations as either reliable (groups 5 and 7) or inaccurate/missing despite prior TOV having been received (groups 1, 4 and 8). A third scenario was also considered, those cases which no payments were recorded, and no COI declaration was provided (group 2). This classification is presented in Table 3.
Analysis of the reliability of conflict of interest) declarations among authorships with records of payments prior to the publication of the clinical practice guideline.
| Classification | n (%) |
|---|---|
| Reliable declaration (groups 5 and 7) | 137 (16.9%) |
| Inaccurate/missing declaration (groups 1, 4 and 8) | 430 (53.1%) |
| Absent declaration and no payment records (group 2) | 57 (7.0%) |
| Not verifiable declaration (groups 3, 6 and 9) | 186 (23.0%) |
| Total | 810 (100%) |
The findings indicate that only 137 (16.9%) of the COI declarations made by authors of the 60 reviewed CPG can be considered reliable, while over half (53.09%, n=430) were either inaccurate or absent.
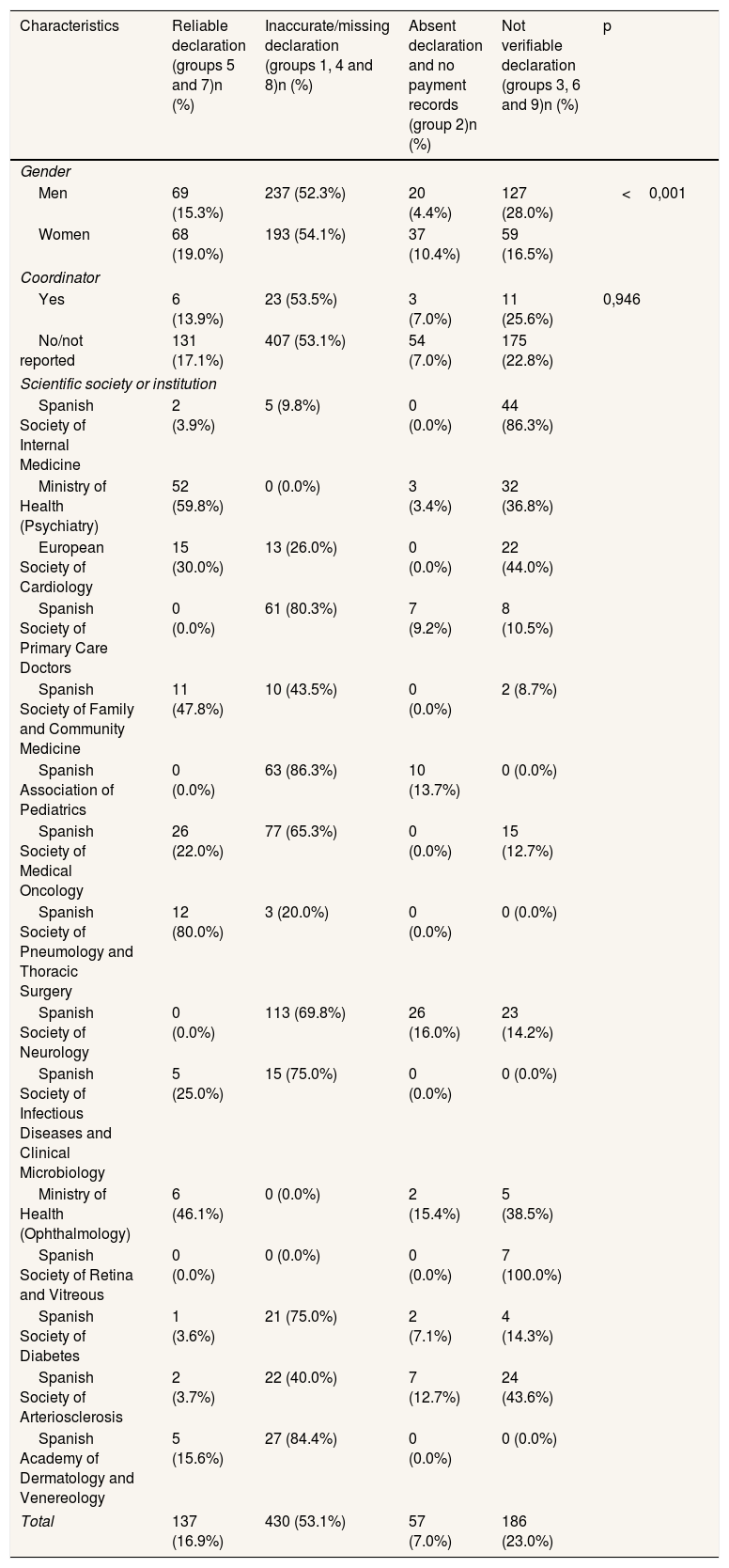
Regarding the reliability of COI declarations by author characteristics, it was observed that, for both men and women, unreliable declarations accounted for more than half of the authorships (52.3 and 54.1%, respectively). Although the frequencies in coordination roles were similar, the reliability of COI declarations was lower among guideline coordinators compared to non-coordinators (13.9% vs. 17.1%). With respect to the scientific societies endorsing the CPGs, five societies were identified in which over 75% of the authorships were associated with unreliable COI declarations. In contrast, only the Ministry of Health and the Spanish Society of Pulmonology and Thoracic Surgery demonstrated over 50% reliability in the declarations submitted (Table 4).
Analysis of conflict of interest declarations for all authorships (n=810), according to gender, coordination role, and the scientific society endorsing the clinical practice guideline.
| Characteristics | Reliable declaration (groups 5 and 7)n (%) | Inaccurate/missing declaration (groups 1, 4 and 8)n (%) | Absent declaration and no payment records (group 2)n (%) | Not verifiable declaration (groups 3, 6 and 9)n (%) | p |
|---|---|---|---|---|---|
| Gender | |||||
| Men | 69 (15.3%) | 237 (52.3%) | 20 (4.4%) | 127 (28.0%) | <0,001 |
| Women | 68 (19.0%) | 193 (54.1%) | 37 (10.4%) | 59 (16.5%) | |
| Coordinator | |||||
| Yes | 6 (13.9%) | 23 (53.5%) | 3 (7.0%) | 11 (25.6%) | 0,946 |
| No/not reported | 131 (17.1%) | 407 (53.1%) | 54 (7.0%) | 175 (22.8%) | |
| Scientific society or institution | |||||
| Spanish Society of Internal Medicine | 2 (3.9%) | 5 (9.8%) | 0 (0.0%) | 44 (86.3%) | |
| Ministry of Health (Psychiatry) | 52 (59.8%) | 0 (0.0%) | 3 (3.4%) | 32 (36.8%) | |
| European Society of Cardiology | 15 (30.0%) | 13 (26.0%) | 0 (0.0%) | 22 (44.0%) | |
| Spanish Society of Primary Care Doctors | 0 (0.0%) | 61 (80.3%) | 7 (9.2%) | 8 (10.5%) | |
| Spanish Society of Family and Community Medicine | 11 (47.8%) | 10 (43.5%) | 0 (0.0%) | 2 (8.7%) | |
| Spanish Association of Pediatrics | 0 (0.0%) | 63 (86.3%) | 10 (13.7%) | 0 (0.0%) | |
| Spanish Society of Medical Oncology | 26 (22.0%) | 77 (65.3%) | 0 (0.0%) | 15 (12.7%) | |
| Spanish Society of Pneumology and Thoracic Surgery | 12 (80.0%) | 3 (20.0%) | 0 (0.0%) | 0 (0.0%) | |
| Spanish Society of Neurology | 0 (0.0%) | 113 (69.8%) | 26 (16.0%) | 23 (14.2%) | |
| Spanish Society of Infectious Diseases and Clinical Microbiology | 5 (25.0%) | 15 (75.0%) | 0 (0.0%) | 0 (0.0%) | |
| Ministry of Health (Ophthalmology) | 6 (46.1%) | 0 (0.0%) | 2 (15.4%) | 5 (38.5%) | |
| Spanish Society of Retina and Vitreous | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 7 (100.0%) | |
| Spanish Society of Diabetes | 1 (3.6%) | 21 (75.0%) | 2 (7.1%) | 4 (14.3%) | |
| Spanish Society of Arteriosclerosis | 2 (3.7%) | 22 (40.0%) | 7 (12.7%) | 24 (43.6%) | |
| Spanish Academy of Dermatology and Venereology | 5 (15.6%) | 27 (84.4%) | 0 (0.0%) | 0 (0.0%) | |
| Total | 137 (16.9%) | 430 (53.1%) | 57 (7.0%) | 186 (23.0%) | |
This study shows a high prevalence of COI among authors in our sample of Spanish CPG, in many cases (46%) COI were not fully disclosed, and the accuracy of COI statements was very low.
Despite recommendations issued by academic and institutional scientific entities regarding transparency in COI declarations within CPG,2,17 our findings indicate that current practices among authors remain inadequate. This persists despite evidence of financial relationships between the professionals involved in scientific evidence production18,19 and the observation that Spanish scientific societies tend to fail to include COI declarations, even when their collaborations with private companies are documented.20
One issue identified was that nearly half (46.42%) of the authorships included no COI declaration. This finding was also noted in tow national based similar studies, in which the study Redeguías and the letter to the editor by Briones et al.21 showed that information relating to financial COI remains scarcer, suggesting that it could reduce both the reliability and the quality of the guidelines.21,22 Furthermore, among the declaration provided, 1 in 10 was incomplete and another 1 in 10 was underreported. These findings are consistent with the evidenced by two recent systematic reviews carried out in 201923 and, as an extension of that work, in 2021,24 which showed that the pooled proportion of discrepancies at the authorship level approached 93%. In addition, pooled proportion of authors who accurately disclosed COIs did not overcome the 18%. In our study, the frequency was approximately 17%.
This issue does not only affect the sample of authorships analysed. Several studies assessing financial COI have shown that such conflicts are often not disclosed. For example, in a survey of 192 authors of 44 CPG, participants reported that in 42 of the guidelines in which they were involved, no declarations related to financial COI were made or required.25 Norris et al.26 found that 40% of 250 CPG did not include authors disclosure information. Despite regulatory changes and recommendation regarding COI reporting, a 2023 review of 197 CGP revealed that 20% still did not report any COI information.27 Similarly, a study carried out by Mooghali et al.28 examining American clinicians’ authorships, found that over 60% of CPG authorships did not submit any COI declaration.
Overall, the evidence consistently suggests that COI are frequently underreported or omitted and agree on the impact that this lack of transparency can have on credibility and confidence.
Although there is existing guidance urging scientific societies that endorse CPG to adopt internal regulatory policies and for participating professionals to minimize their conflicts in practice,2 COI declaration within CPG endorsed by scientific societies remain limited. In our analysis, only some institutions such as the Ministry of Health did not include unreliable COI disclosures.
The relationship between pharmaceutical and other private interest and scientific societies is evident, largely due to the disparity in funding capacity between public institutions and private biomedical interest.29,30 In Spain, the situation appears similar, transparency regarding COI declarations remains limited. Although several studies have addressed this issue,21,31–33 formal declarations are still uncommon, and some prominent scientific societies continue to lack comprehensive COI transparency.
These findings underscore not only the need for clearer guidance to declare potential COI but also highlight the importance of applying ethical frameworks, such as the GIN principles,34 and evaluation tools, such as the AGREE framework or IGEST, for all guidelines to ensure the clinical quality, integrity, and independence of CPG.
The interaction between pharmaceutical industry, health professionals, and science societies within healthcare ecosystem, and in general the existence of financial bias is an area of growing concern, as such interactions has been associated with questionable prescribing behaviours by health professionals.35 A systematic review conducted by Mitchell et al.36 demonstrated how these interactions influence and undermine the independent therapeutic practice of clinicians. Reducing the financial bias from the institutions, scientific societies and CPGs remains a priority.37
LimitationsThis study has several limitations. Although our selection of professionals and guidelines was comprehensive, it captures only the COI disclosure landscape of individually selected CPG related with some major causes of disease burden in Spain published in 2016,13 potentially omitting relevant guidelines addressing prevalent health issues with undisclosed conflicts. The sample of authors was limited to those involved in nationally applicable guidelines or those endorsed by Spanish scientific societies, excluding other scientific roles. Another limitation was that gender was inferred from full names, introducing possible misclassification in the absence of self-identification.
A relevant limitation during data extraction in this investigation was the public TOV records availability. While some companies publish accessible documents, others limit their publication to the last three years before the actual date, or they provide documents in non-editable formats (images), which prevented automated data processing and required manual identification and extraction of the data. This creates a heterogeneity in our records selection, which is inevitable since we do not count on a centralized COI database such as OpenPayments in USA. Another limitation is the matching process between the authorship disclosure and the industry records, only referenced by full names and not with an accurate identifier, what could have improved our capability to detect every author's name in the documents and subsequently identify more TOV. Nonetheless, our results confirm that a more sensitive selection would only involve lower reliable disclosures rates, as improving all these limitations would only detect higher rates of TOV from authors with very low transparency attitudes.
Accordingly, we experienced a temporal heterogeneity due to our records availability, which makes newer guideline authors more likely to have TOV detected during our investigation, as a bigger number of previous records were studied.
The analysis relied on aggregated TOV from pharmaceutical companies, without differentiating types of payments. Moreover, some CPG focused on conditions managed primarily through surgical or non-pharmacological interventions, which may have affected COI assessment. Finally, the selected companies do not fully represent industry influence but allow us to evaluate properly CPG authors’ COI declaration. Future studies should broaden the scope to include both pharmaceutical and non-pharmaceutical companies, widely selected, to improve COI detection.
ConclusionsThis study demonstrates a lack of adequate COI disclosure among Spanish authors of CPG. Considering the potential influence that clinical recommendations provided in guidelines can exert on healthcare professionals’ decision-making and therapeutic prescription, proper, transparent and regularly updated COI disclosures are essential to ensure the independence and integrity of scientific recommendations. Further research is needed to determine the effect of non-declared COI on clinical recommendations and healthcare decision-making.
Some measures seem to be beneficial to safeguard scientific production integrity and transparency. Scientific societies and professionals should strengthen ethical oversight not only in the development of CPG, but also in all areas where expert knowledge is needed. The current system allows individuals to conceal and not declare COIs without facing any real consequences, thereby undermining trust in scientific outputs.
It would be therefore helpful to implement platforms that facilitate the identification of COI among healthcare professionals and scientists, as other countries established. All stakeholders in health ecosystem should commit to providing transparent, reliable and accessible disclosures.
Availability of databases and material for replicationThe anonymized database used in this study can be consulted via Mendeley Data, in the following link: https://data.mendeley.com/datasets/xgbhfs629f/1
Conflicts of interest in clinical practice guidelines remain a significant limitation due to underreported transfers of value. In Spain, limited evidence regarding transparency raises concerns about the Independence of these clinical recommendations.
What does this study add to literature?This study describes conflicts of interest among Spanish guideline authors, highlighting that 80% received payments, yet only 17% of declarations were reliable.
What are the implications of the results?Institutions that produce clinical guidelines must establish measures to reinforce ethics, independence and trust. Governments must establish regulations to prevent guidelines without guarantees of independence from being applied in healthcare; and require pharmaceutical companies to implement a single system of maximum transparency for declaring payments to healthcare professionals and their associations.
Ferrán Catalá-López.
Transparency declarationThe corresponding author, on behalf of the other authors guarantee the accuracy, transparency and honesty of the data and information contained in the study, that no relevant information has been omitted and that all discrepancies between authors have been adequately resolved and described.
Authorship contributionsI. Hernández-Aguado, S. Martín-Benlloch and S. Morales-Garzón conceived and designed the study. S. Martín-Benlloch, in collaboration with A.M. Martín Fernández-Gallardo, developed the data collection, and S. Martín-Benlloch conducted the data curation and analysis, with methodological support from S. Morales-Garzón. All authors contributed to the interpretation of the results. S. Martín-Benlloch and S. Morales-Garzón drafted the first version of the manuscript. I. Hernández-Aguado and A.M. Martín Fernández-Gallardo critically revised the manuscript. I. Hernández-Aguado provided senior oversight and ensured the quality and integrity of the work. All authors approved the final version for publication and agree to be accountable for all aspects of the work.
FundingNone.
Conflicts of InterestNone.